Education
HCOOCH CH2 H2O Reaction Explained for Students
Chemistry often involves studying how different compounds interact with one another. Learning about reactions helps students understand how bonds form, break, and rearrange to produce new products. One reaction that can be discussed in this context is HCOOCH CH2 H2O. While at first this may look confusing, breaking it down step by step makes it easier to follow. This article explains the reaction, the structure of the compounds involved, and their importance for students studying organic chemistry.
Understanding the Formula
The formula HCOOCH CH2 H2O looks unusual because it combines different fragments of chemical notation. To understand it, let us look at the components individually.
- HCOO refers to a formyl or formate group, which is linked to formic acid derivatives.
- CH2 indicates a carbon group with two hydrogen atoms attached, which is often part of larger organic structures.
- H2O represents water, a key reactant in many organic reactions such as hydrolysis.
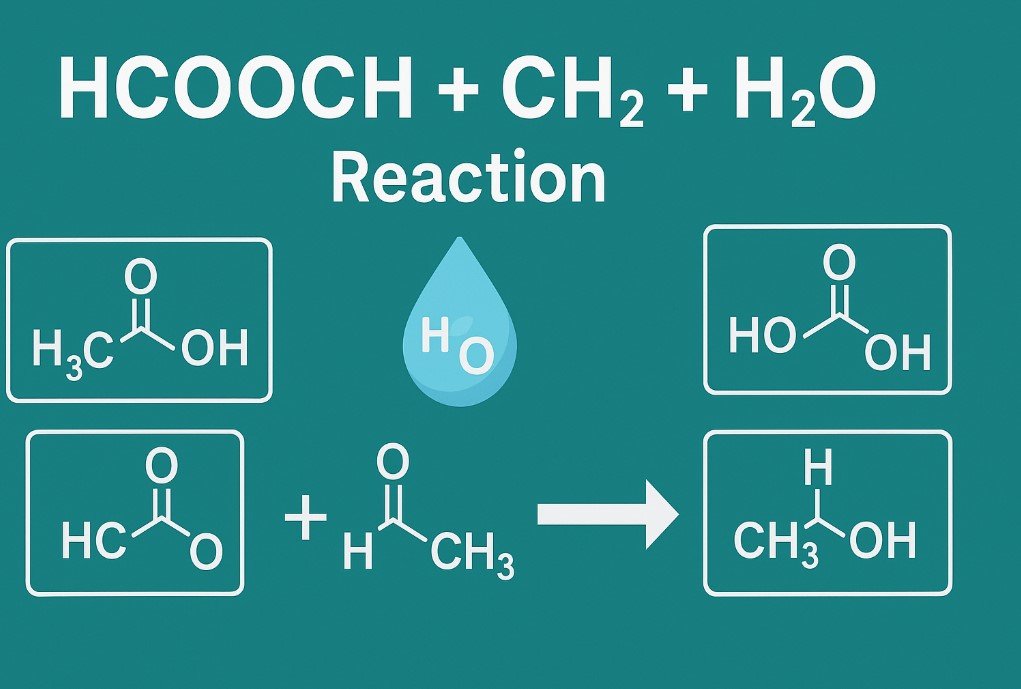
When you put these together, you are dealing with a reaction where a formate ester or related compound reacts with water in the presence of a CH2 group. This type of process usually leads to hydrolysis, breaking an ester bond to produce an alcohol and an acid.
The Reaction Process
In general, when an ester reacts with water under the right conditions, the reaction is called hydrolysis. Hydrolysis means “splitting with water.” The ester bond is broken, and the result is the formation of an acid and an alcohol. In this case, HCOOCH CH2 H2O is showing how water reacts with a formate ester that includes a CH2 group.
The key steps in the hydrolysis process are:
- Water molecules attack the carbon atom connected to the oxygen in the ester group.
- The bond between carbon and oxygen in the ester is broken.
- New compounds are formed: one part becomes formic acid (HCOOH), and the other part becomes an alcohol that includes the CH2 group.
This type of reaction is commonly studied in organic chemistry because it demonstrates the behavior of esters in the presence of water.
Why Water Is Important
Water plays a major role in the reaction HCOOCH CH2 H2O. Without water, hydrolysis cannot occur. Water acts as a reactant that supplies both a hydrogen atom and a hydroxyl group. Together, these attach to different parts of the broken ester, leading to the final products. This shows why water is not only a solvent in many cases but also a reactant that changes the outcome of the process.
Products of the Reaction
When an ester such as HCOOCH reacts with water, the products depend on the structure of the ester. For HCOOCH CH2 H2O, the likely products are formic acid (HCOOH) and a simple alcohol containing the CH2 group, such as methanol or a related compound.
Formic acid is the simplest carboxylic acid and has uses in biology and industry. Methanol is a basic alcohol with wide applications as a solvent and fuel component. These products demonstrate how even small reactions can lead to compounds that are important in real-world settings.
Relevance for Students
For students, studying reactions like HCOOCH CH2 H2O provides important lessons:
- It teaches how to identify functional groups such as esters.
- It shows how hydrolysis reactions work with the help of water.
- It connects theory to practical products like acids and alcohols.
Understanding these reactions also prepares students for advanced chemistry topics where more complex esters, proteins, or lipids undergo hydrolysis in biological systems.
Applications in Real Life
Although HCOOCH CH2 H2O looks like a simple reaction on paper, the hydrolysis of esters has wide applications. Some examples include:
- Breaking down fats and oils into glycerol and fatty acids in food chemistry
- Producing alcohols for industrial use
- Creating carboxylic acids that are used in pharmaceuticals and chemical production
- Explaining biological processes such as the digestion of lipids in humans
By studying one example reaction, students can see how the concept applies to many other important processes.
Learning Strategies
When approaching reactions such as HCOOCH CH2 H2O, students should focus on a few key strategies:
- Identify the functional groups involved, such as esters, acids, or alcohols.
- Practice writing balanced chemical equations for hydrolysis.
- Connect the reaction to real-life examples in food, biology, or industry.
- Use visual diagrams of molecules to understand how bonds break and form.
These strategies make it easier to understand not only this reaction but also others that follow similar mechanisms.
Challenges Students Face
One challenge is recognizing that the formula HCOOCH CH2 H2O is not written in the same way as typical balanced equations. Students may struggle to see how the parts fit together. Another challenge is understanding the mechanism of hydrolysis, where water directly participates in breaking a bond. With practice, these difficulties become easier to manage. Teachers often recommend drawing the step-by-step mechanism to clarify how atoms move during the reaction.
Summary of Key Points
To make the explanation clearer, here are the main ideas about the reaction:
- HCOOCH CH2 H2O represents a hydrolysis process involving a formate ester and water.
- The reaction breaks the ester bond, producing formic acid and an alcohol.
- Water acts as the key reactant, not just as a solvent.
- The reaction is an example of how esters behave in organic chemistry.
- Understanding it helps students connect classroom learning with real-life chemistry.
Conclusion
The reaction expressed as HCOOCH CH2 H2O is an example of ester hydrolysis, where water breaks down a compound into an acid and an alcohol. For students, it illustrates important ideas about functional groups, the role of water in organic chemistry, and the production of useful products. Although the notation may look complicated at first, breaking it into smaller parts reveals a straightforward reaction. Studying processes like this gives students both theoretical knowledge and practical understanding, helping them prepare for advanced learning in chemistry and real-world applications.











